The atoms were unchangeable, indestructible, and always existed. He taught that there were substances called atoms and that these atoms made up all material things. Quarks are elementary because quarks cannot be broken down any further. One of these philosophers was Democritus (460-370 B.C.E.), often referred to as the 'laughing philosopher' because of his emphasis on cheerfulness.
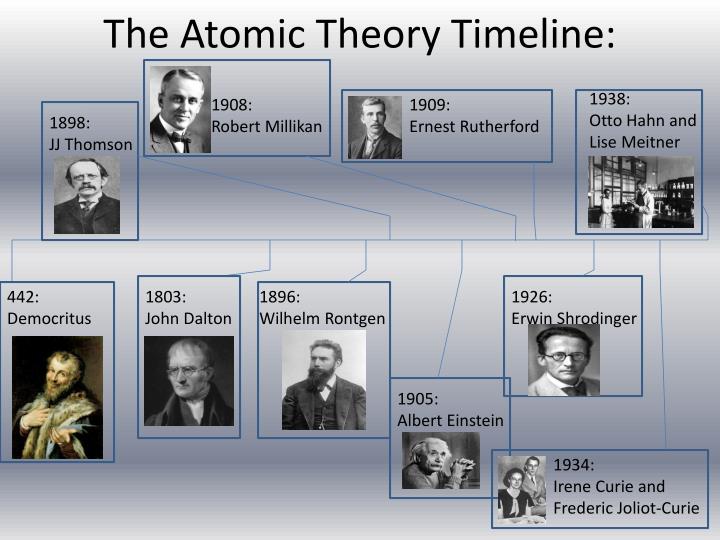
Protons and neutrons are also not elementary particles because they are made up of even smaller particles called quarks joined together by other particles called gluons (because they "glue" the quarks together in the atom). Modern physicsĪtoms are not elementary particles, because they are made of subatomic particles like protons and neutrons. Define isotopes and give examples for several elements. Describe the three subatomic particles that compose atoms. Summarize and interpret the results of the experiments of Thomson, Millikan, and Rutherford.

Some idea of present-day atomic physics can be found in the links in the table below. By the end of this section, you will be able to: Outline milestones in the development of modern atomic theory. Although this model was well understood, modern physics has developed further, and present-day ideas cannot be made easy to understand. Isotopes vary in the number of neutrons present in the nucleus. 2.2: Development of Modern Atomic Tehory is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. This experiment was called the Geiger–Marsden experiment or the Gold Foil Experimentīy this stage the main elements of the atom were clear, plus the discovery that atoms of an element may occur in isotopes. Three important kinds of radiation are particles (helium nuclei), particles (electrons traveling at high speed), and rays (similar to x-rays but higher in energy). Rutherford showed this when he used an alpha radiation source (from helium) to hit the very thin gold sheets, surrounded by a Zinc sulphide lampshade that produced visible light when hit by alpha emissions. In 1910, the New Zealand physicist Ernest Rutherford put forward the idea that the positive charges of the atom were found mostly in its center, in the nucleus, and the electrons ( e-) around it. He proposed that electrons are arranged in concentric circular orbits around the nucleus. Lord Ernest Rutherford later named these positively charged particles protons Rutherford's atomic modelĪtomic experiment of Lord Ernest Rutherford In 1913, Neils Bohr, a student of Rutherford s, developed a new model of the atom.
In the same time, experiments by Eugene Goldstein in 1886 with cathode discharge tubes allowed him to establish that the positive charges had a mass of 1.6726 * 10 −27 kg and an electrical charge of +1,6 * 10 −19 C. In 1906, Robert Millikan determined that the electrons had a Coulomb (C) charge of -1.6 * 10 −19, something that allowed calculation of its mass as tiny, equal to 9.109 * 10 −31 kg. The negative charges were named electrons ( e-).Īccording to the assumptions established about the atoms neutral charge, Thomson proposed the first atomic model, that was described as a positively-charged sphere in which the electrons were inlaid (with negative charges). Thomson knew that the atoms were electrically neutral, but he established that, for this to occur, an atom should have the same quantity of negative and positive charges. Crookes named the emission ' cathode rays'.Īfter the cathode ray experiments, Sir Joseph John Thomson established that the emitted ray was formed by negative charges, because they were attracted by the positive pole. When creating a vacuum in the tube, a light discharge can be seen that goes from the cathode (negatively-charged electrode) to the anode (positively-charged electrode). (c) In the cathode ray, the beam (shown in yellow) comes from the. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube.

In 1850, Sir William Crookes constructed a ' discharge tube', that is a glass tube with the air removed and metallic electrodes at its ends, connected to a high voltage source. The results of these measurements indicated that these particles were much lighter than atoms (Figure 3.3.1 3.3. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4 ), is a fundamental concept that states that all elements are composed of atoms.
